Source: Cornell University
By: Lorianna Keuhn and Tyler Olson
As the field of genetics has advanced, new undesirable haplotypes have been discovered. Among these are fertility haplotypes and congenital defects. As genotyping has become more prevalent on farms, the question of what to do with carriers of these haplotypes has arisen. While some may believe that these animals should just be culled, there are also other options available such as embryo transfer recipients or more careful breeding. Haplotypes which result in embryonic loss when combined, are typically calculated into the DPR (Daughter Pregnancy Rate), SCR (Sire Conception Rate), and LNM (Lifetime Net Merit) for that bull, and may not be listed separately.
Below is information about 7 of the haplotypes that have been discovered since the development of genetics as well as strategies to manage these and other similar diseases.
Complex Vertebral Malformation (CVM)
Origin: CVM was first discovered in the Holstein breed in 1999 and has since been discovered in the Danish, United States, United Kingdom, Netherlands, and Japanese herds. Analyzation of pedigree records traced the disorder to a single common bull. This bull was Carlin-M Ivanhoe Bell who was used widely for many years. This wide usage of one bull has caused a high percentage of carriers in herds, 31% in Denmark and 32.5% in Japan.
Mode of Inheritance: Autosomal recessive
Phenotype: There have been multiple phenotypes associated with the disease including hemivertebrae, misshaped vertebrae, ankylosis of cervico-thoracic vertebrae, scoliosis, and more. Abortion can also result from the disease. Only animals that are homozygous for the mutation are affected. There has been no symptoms of the disease seen in carriers thus far.
Pros: Carlin-M Ivanhoe Bell was used so much for breeding because his daughters had high milk production. High milk production has been associated with carriers of this disease.
Test: Because there are so many phenotypes a genetic test is the only sure diagnosis.
Bovine Brachyspina Syndrome
Origin: Pedigree information on all cases leads back to Sweet Haven Tradition who was a common bull used in the late 70s and 80s. Carrier frequency is estimated to be between 6 and 7% in the Holstein breed.
Mode of Inheritance: Autosomal recessive
Phenotype: The main phenotype seen is brachyspina or significant shortening of the spine. Other phenotypes include growth retardation, extremely reduced body weight, uneven alignment of the upper and lower teeth, and malformation of inner organs. Abortion is also a possibility. There have been no symptoms seen in carriers thus far.
Test: Registered Holstein bulls have been reported as carriers (BY) or tested non-carriers (TY) for Brachyspina
Bovine Leukocyte Adhesion Deficiency (BLAD)
Origin: Pedigree information can trace the disease back to Osborndale Ivanhoe. He was a common bull used in the mid-50s and 60s.
Mode of Inheritance: Autosomal Recessive
Phenotype: Animals with BLAD have recurring infections of soft tissues, fever, low appetite, chronic pneumonia, and diarrhea. They also have severe ulcers, stunted growth, and impaired wound healing.
Fertility Haplotypes
Origin: There is little know about the genetics of this disease because affected individuals are generally aborted. Because of this they have simply been named Holstein Haplotype 1-5 (HH1, HH2, HH3, HH4, HH5), Jersey Haplotype 1 (JH1), Brown Swiss Haplotype 1 and 2 (BH1 and BH2), and Ayrshire Haplotype 1 (AH1).
Mode of Inheritance: Autosomal recessive
Phenotype: When two carriers are bred the homozygous embryo is generally aborted. Each haplotype has a normal time of pregnancy loss. Pregnancy can be terminated at all stages for HH1, before day 100 for HH2 and BH1, before day 60 for HH3, HH5, and JH1. It is unknown when the pregnancy is terminated for HH4 and AH1. BH2 produces stillborn animals. It is important to note that haplotypes that cause abortion later in life have a higher economic impact.
Test: Animals that are carriers are listed with the specific haplotype they have followed by a C, for example HH1C. Animals that are not carriers are labeled with the haplotype followed by a T, for example HH1T.
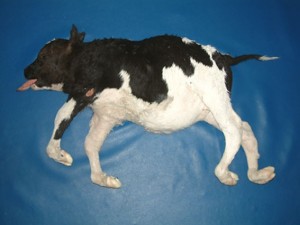
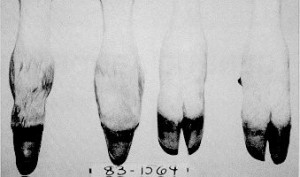
Syndactyly (Mule Foot)
Origin: Syndactyly has been reported in multiple breeds including Holstein, Aberdeen–Angus, Simmental, Brown Swiss, Chianina, Japanese Native, Hariana, Swedish Red Pied, and Czech Black Pied. Has been traced back to a single ancestor in the Holstein.
Mode of Inheritance: Autosomal Recessive with variable penetrance
Phenotype: Animals have a fusion of the two digits, causing the animal to have only one claw on at least one foot. In Holsteins animals that are carries can partially show the disease. The front feet are most commonly affected.
Pros: Carriers are associated with superior milk and butterfat production.
Weaver
Origin: So far only found in the Brown Swiss it is believed to be a mutation that occurred during a mating in the 1920s or 30s that was a result of inbreeding. The technical name is Bovine Progressive Degenerative Myelo-encephalopathy but because the animals have a weaving gait the name “Weaver” developed.
Mode of Inheritance: Autosomal Recessive
Phenotype: Animals show bilateral hind leg weakness and inability to coordinate movements between the age of 6-18 months resulting in a weaving gait. They have abnormal reflexes and function in the hind legs but they have normal motor and sensory reflexes. They also don’t have any musculoskeletal abnormalities.
Pros: There has been an association between carrier status and production. Animals that are carriers produce 690kg more milk and 26.2 kg more butterfat on average.
Haplotype for Cholesterol Deficiency (HCD)
Origin: HCD was recently discovered in 2015 by German scientist. It have been traced back to Maughlin Storm who is the last common genotyped bull that possesses the haplotype. The exact defect has not yet been identified but a region contain the haplotype has been determined.
Mode of Inheritance: Autosomal recessive
Phenotype: Animals have trouble metabolizing cholesterol. This causes weight loss, low appetite, weakness, and scours that do not respond to treatment. Animals typically live about 6 months.
Test: It is difficult to test if the animal has the undesirable mutation. A close relative to Storm was Mark Anthony who had a similar but normal haplotype. For this reason in order to determine if an animal is actually affected they must be tested and have their pedigree analyzed. If they test positive but they cannot confirm that Storm is in the pedigree then the animal is listed as a potential carrier. Because of this a scale has been created for animals to be scored on.
| Haplotype Code | % Affected Animals | Description |
| 0 | 94% | Non-carrier: free of HCD |
| 1 | 4.4% | Carrier: confirmed on both sides of pedigree |
| 2 | 0.03% | Homozygous: confirmed on both sides of pedigree |
| 3 | 1.6% | Suspect carrier: haplotype origin could not be confirmed to pedigree |
| 4 | 0.02% | Suspect homozygous: Probable carrier and may be homozygous; origin of haplotypes could not be confirmed |
Economic Impact:
Producing affected animals is costly to the farm. Diseases that have early abortions are the least costly because you are spending less resources on these animals. If you are wanting to keep carrier cows in the herd still they can be bred back quicker as well. For animals that are still born more resources are used during pregnancy and the dams can not be bred back as fast. For diseases such as HCD and weaver where the animal is born alive and does not tend to show signs for a little even more resources are used on the animal. All of these diseases lead to economic loss but they vary in the amount of loss.
Testing:
There are a few ways that you can get your animals tested for these diseases. The two major genotyping companies right now are Geneseek and Zoetis. Both offer genomic evaluations for dairy and beef cattle. There are a few different levels of evaluations that are based on the number of markers being used in the test and how much information you get back. Both companies also offer add-ons to the tests giving you back more information. (For more information on the genotyping process see PAGE) You can also get testing information from breed associations.
Geneseek offers a specific profiler test called the Geneseek Genomic Profiler or GGP. This test includes multiple genetics conditions including CVM, BLAD, Brachyspina, HH1, HH2, HH3, and JH1. They also offer tests for syndactyly and HCD.
Zoetis offers a Dairy Wellness Profit Index which provides fertility haplotypes, CVM, and brachyspina testing.
Disease Control:
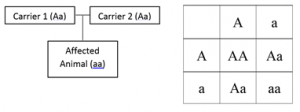
All of the above mentioned diseases are autosomal recessive meaning that two carriers of the disease must be bred together to obtain an animal with a disease. Even when two carriers are bred together a homozygous animal is only produced 25% of the time.
In this figure two carriers were bred together to produce a homozygous recessive animal. Each parent had one normal allele, A, and one disease allele, a. You can predict the likelihood of the offspring’s genotype using a punnet square (right side of figure). When two carriers are bred you are likely to get a non-carrier animal (AA) 25% of the time, another carrier animal (Aa) 50% of the time, and an affected animal (aa) 25% of the time.
If you would like to keep carrier animals in your herd you must make sure that carrier animals are never bred together. If you have an animal that is tested and proven to be a carrier or has a carrier animal in its pedigree then you should be sure to not breed that animal to a carrier bull. Almost all bulls used in AI are tested for these diseases and the results are available through the breed association.
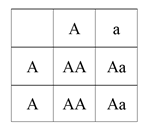
There are a few ways to manage these diseases. If your farm is genetically testing all of its animals you can also test for these diseases. You can look at pedigree records and determine if any of the animals in the pedigree, specifically bulls because almost all AI bulls are genotyped, are carriers of the disease. Having all of this information is best but if you only have one you can still make better mating decisions. Once you know this information you need to decide if you want to eliminate the disease from your herd or if you would like to keep carrier animals. For some of the diseases such as CVM, Syndactyly, and Weaver carriers are associated with higher milk production, and so you may want to have carriers in your herd. For other diseases like fertility haplotypes, HCD, BLAD, and Brachyspina there is not apparent advantage to having carriers in your herd meaning you may want to breed this disease out. If you have a carrier cow and mate it to a non-carrier bull you will produce a carrier animal (Aa) 50% of the time and a non-carrier animal (AA) the other 50% of the time. This can be seen in the figure below.
If you would like to breed out the disease there are a few ways that it can be done. You can either breed the animals to a terminal beef sire and continue to milk them. You could use them as embryo recipients for embryo transfer. Culling all the animals is an option but it is more cost effective to keep them producing in the herd until they are older and just not breed them for replacements.
You can find a few of the above mentioned diseases in the Genetic Disease Brochure as well.
References
BUILD A HEALTHIER HERD THROUGH BETTER GENETICS. (n.d.). Retrieved May 16, 2016, from https://www.zoetisus.com/animal-genetics/dairy/clarifide/clarifide-plus.aspx
Drögemüller, C., & Distl, O. (2006). Genetic analysis of syndactyly in German Holstein cattle. The Veterinary Journal, 171(1), 120-125.
Duchesne, A., Gautier, M., Chadi, S., Grohs, C., Floriot, S., Gallard, Y., … & Eggen, A. (2006). Identification of a doublet missense substitution in the bovine LRP4 gene as a candidate causal mutation for syndactyly in Holstein cattle. Genomics, 88(5), 610-621.
Frequently Asked Questions About Brachyspina. (n.d.). Retrieved May 15, 2016, from http://www.holsteinusa.com/pdf/brachyspina_faq.pdf
Frequently Asked Questions About Complex Vertebral Malformation (CVM) Retrieved May 15, 2016, from http://www.selectsires.com/resources/healthdocs/cvm.html?version=20160420
GENETIC ABNORMALITIES IN BROWN SWISS CATTLE. (n.d.). Retrieved May 15, 2016, from http://www.brownswissusa.com/Portals/0/Documents/Abnormality Pamphlet.pdf
Georges, Michel et al. Microsatellite mapping of the gene causing weaver disease in cattle will allow the study of an associated quantitative trait locus.
Genomic Solutions for Dairy Cattle. (n.d.). Retrieved May 16, 2016, from http://www.neogen.com/Genomics/Dairy.html
Haplotypes Impacting Fertility. (n.d.). Retrieved May 15, 2016, from http://www.selectsires.com/resources/healthdocs/impactingfertility.html?version=201604
NAGAHATA, H. (2004). Bovine leukocyte adhesion deficiency (BLAD): a review. Journal of Veterinary Medical Science, 66(12), 1475-1482.
Syndactyly (Mule Foot). (n.d.). Retrieved May 15, 2016, from http://calfology.com/library/wiki/syndactyly-mule-foot
Thomsen B., Horn P., Panitz F., Bendixen E., Peterson A., Holm LE. 2006. A missense mutation in the bovine SCL35A3 gene, encoding a UDP-N-acetylglucosamine transporter, causes complex vertebral malformation. Genome Research, 16(1), 97-105.
Update on Holstein Haplotype associated with Cholesterol Deficiency (HCD) for August Genetic Evaluations. (n.d.). Retrieved May 15, 2016, from https://www.zoetisus.com/animal-genetics/media/documents/press-releases/update-on-holstein-haplotype-for-cholesterol-deficiency-_hcd_.pdf